 
Zircon mixed with praseodymium (Pr) or vanadium (V) makes blue and yellow pigments for glazing pottery.Ĭubic zirconia (crystalline form of zirconium dioxide) is a synthetic gemstone, The colourless stones, when cut, resemble diamonds. Zircon (ZrSiO 4) is a natural semi-precious gemstone and has a high index of refraction that found in a variety of colours. Zirconium is used extensively by the chemical industry where metal is protected by a thin oxide layer, making it exceptionally resistant to corrosion by acids, alkalis and seawater. With Niobium, zirconium is superconductive at low temperatures and is used to make superconductive magnets, which offer direct large-scale generation of electric power. Some Nuclear reactors have more than 100,000 metres of zirconium alloy tubing. It is an ideal material for use in nuclear power station, where it is used more than 90% in this way. Zirconium used in alloys such as Zircaloy ( 98% Zr & 2% Sn, Fe, Cr, Nb, Ni), which is used in Nuclear energy application because of it does not absorb neutrons. Zirconium is used in removal of residual gases from vacuum tubes, and as an alloying agent in steel, in surgical instruments, photographic flash bulbs, rayon spinnerets, explosive primers, lamp filaments, etc. Naming: After zircon, zargun (meaning “Gold-colored)ĭiscovery: Martin Heinrich Klaproth (1789)įirst isolation:Jons Jakob Berzelius (1824) Zirconium Uses Zr (s) + 2 I 2 (g) → ZrI 4 (s) (zirconium (IV) iodide) Zirconium History Zr (s) + 2 Br 2 (g) → ZrBr 4 (s) (zirconium (IV) bromide) Zr (s) + 2 Cl 2 (g) → ZrCl 4 (s) (zirconium (IV) chloride) Zr (s) + 2 F 2 (g) → ZrF 4 (s) (Zirconium (IV) fluoride) React with Halogens when heated to form zirconium (IV) halides: It reacts with water at high temperatures, producing hydrogen gas: Zirconium doesn’t react with air under normal condition, But If burn with oxygen than It form: Isotopes: 88 zr 89 Zr 90 Zr 91 Zr 92 Zr 93 Zr 94 Zr 96 Zr Isotope  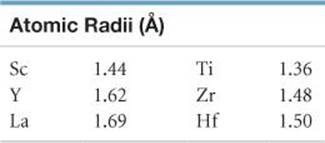
Hexagonal Close Packed (HCP) Reactivity of ZirconiumĮlectron affinity: 41.1 kJ/mol Nuclear Properties of Zirconium Ionization energies: 1st: 640.1 kJ.mol 2nd: 1270 kJ/mol 3rd: 2218 kJ/molĬrystal structure: Hexagonal close-packed Sound Speed: 3800 m/s Atomic Properties of Zirconium Molar magnetic susceptibility: 1.53×10 -9 m 3/mol Physical Properties of Zirconiumĭensity: 6.52 g/cm 3 (In solid) 5.8 g/cm 3 (In Liquid) Mass magnetic susceptibility: 16.8×10 -9 m 3/kg Thermal conductivity: 22.6 W/(m∙K) Electrical properties of ZirconiumĬritical point (Superconducting point): 0.61 K (-272.54 oC, -458.57 oF) Magnetic Properties of Zirconium Zirconium Electron Configuration Thermal Properties of Zirconiumĭebye temperature: 291 K (17.85 oC, 64.13 oF) 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
